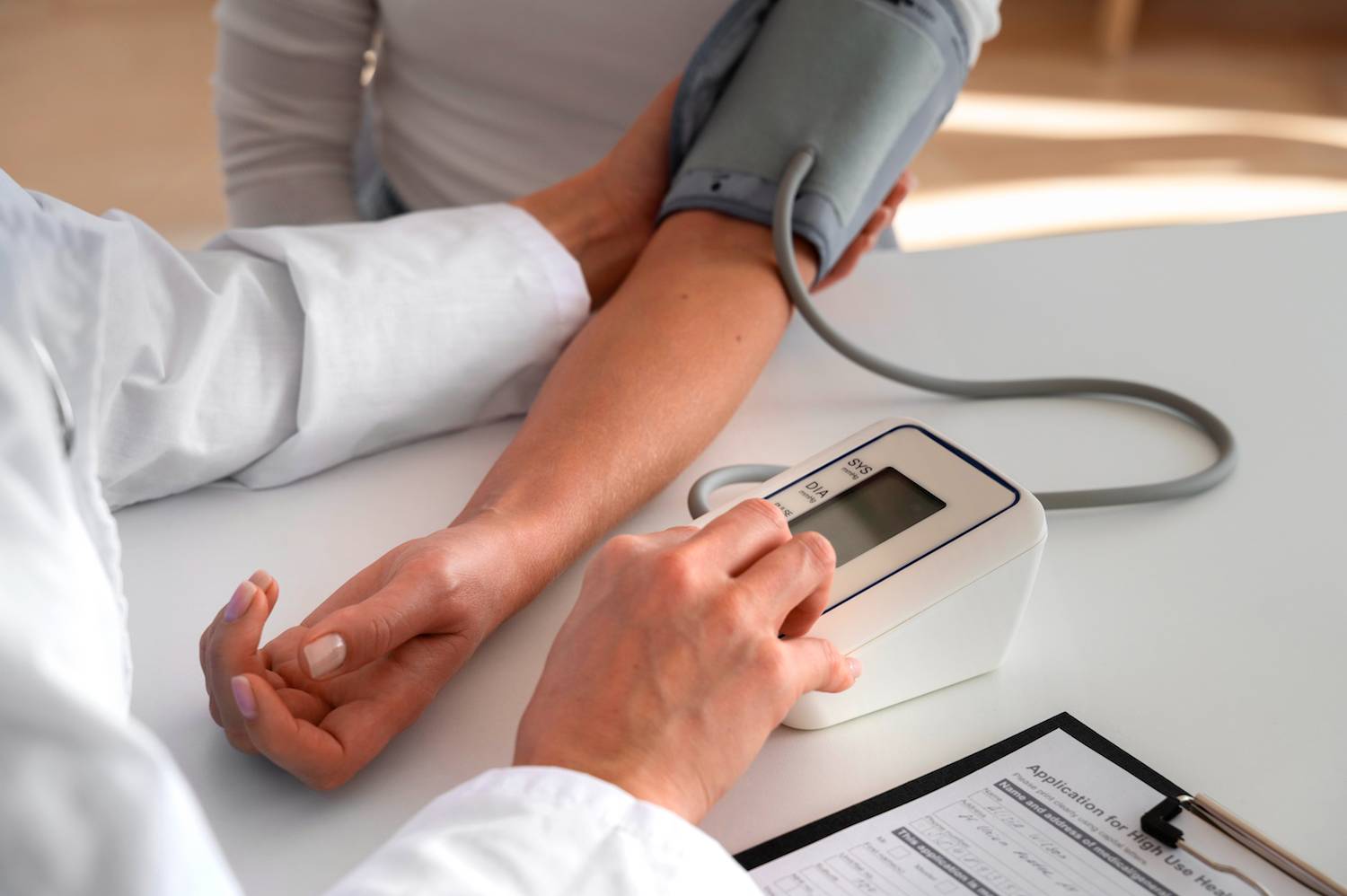
FDA Clears Nanowear's SimpleSense Non-Invasive Continuous Blood Pressure Monitor

FDA Clears Nanowear's SimpleSense Non-Invasive Continuous Blood Pressure Monitor
Nanowear's remote monitoring device and its SimpleSense platform received FDA 510(k) clearance as a continuous blood pressure monitor.

Nanowear Receives FDA Approval for SimpleSense Continuous Blood

NAMSA: Customized biostatistics programs for medical devices

Commercial Partnership — Nanowear

Nanowear SimpleSense Wearable Receives FDA Clearance

Inside Precision Medicine (@Inside_PM) / X

FDA 510(k) Clearance — Nanowear

AliveCor Collaborates With AstraZeneca to Develop Noninvasive

Tensys Real-Time, Noninvasive Blood Pressure Monitor

FDA Approves CareTaker® Wireless Remote Patient Monitor For

AdvaMed (@AdvaMedUpdate) / X

AdvaMed (@AdvaMedUpdate) / X

Nanowear's SimpleSense-BP Cleared for Blood Pressure Monitoring